PhD position: Come and work with us!
If you are thinking about doing a PhD and think that bioinformatics and antibiotic resistance is a cool subject, then now is your chance to come and join us for the next four years! There is a PhD position open i Joakim Larsson’s group, which means that if you get the job you will work with me, Joakim Larsson, Erik Kristiansson, Ørjan Samuelsen and Carl-Fredrik Flach on a super-interesting project relating to discovery of novel beta-lactamase genes (NoCURE). The project aims to better understand where, how and under what circumstances these genetic transfer events take place, in order to provide opportunities to limit or delay resistance development and thus increase the functional lifespan of precious antibiotics. The lion’s share of the work will be related to interpreting large-scale sequencing data generated by collaborators within the project; both genome sequencing and metagenomic data.
This is a great opportunity to prove your bioinformatics skills and use them for something urgently important. Full details about the position can be found here.
Metaxa2 is here!
The new version of Metaxa – Metaxa2 – which I first started talking about more than 1.5 years ago, has finally been determined to be so stable that we can officially release it! The release come around the same time as we submitted a paper describing the changes in it, but I will briefly go through the changes here:
- Metaxa2 now handles extraction and classification of LSU rRNA sequences in addition to SSU rRNA
- The classification engine has been completely redesigned, and now enables accurate taxonomic classifications down to the genus – or in some cases – species level
- The classification database has been updated, and is now based on the SILVA 111 release
- The Metaxa2 Taxonomic Traversal Tool – metaxa2_ttt – has been added to the package, to ease the counting of rRNA sequences in different organism groups (at various taxonomic levels)
- Metaxa2 adds support for paired-end libraries
- It is now possible to directly input of sequences in FASTQ-format to Metaxa2
- The support for libraries with short read lengths (~100 bp) has been vastly improved (and is now assumed to be the case for default settings)
- Metaxa2 can do quality pre-filtering of reads in FASTQ-format
- Metaxa2 adds support for the modern BLAST+ package (although the old blastall version is still default)
- Compatibility with the HMMER 3.1 beta
Metaxa2 brings together a large set of features that we have been gradually incorporating since 2011, many of which have been dependent on each other. Most of the new features and changes are thoroughly explained in the manual. While we hope Metaxa2 is bug free, there will likely be bugs caused by usage scenarios we have not envisioned. I therefore encourage anyone who come across some unexpected behavior to send me an e-mail. Especially, I would like to know about how the software performs using HMMER 3.1 and BLAST+, where testing has been limited compared to older parts of the code.
We hope that you will find Metaxa2 useful, and that it will bring taxonomic assessment of metagenomes another step forward! Metaxa2 can be downloaded here.
New year – Some updates
A new year has begun, and it brings with it a few updates on the website. I have added a summary of the year 2013 from my perspective, and (as you may recognize) updated my picture on the front page. Briefly, this year will bring lots of exciting stuff. Personally, I am quite excited to finally be able to share the new version of Metaxa – Metaxa2 – which will be released to the public late this Winter (or early Spring). Additionally, I look forward to wrap up some manuscript on metagenomics and antibiotic resistance, which I have been working with for more than 2.5 years now. Also, we look forward to some super-intersting technology developments in DNA sequencing, with PacBio finally finding proper usage scenarios, Nano-pore sequencing around the corner, and super-multiplexing on the Illumina instruments. We’re in for a treat with DNA sequencing in 2014!
Talk tomorrow at Swedish Bioinformatics Workshop
Those of you attending the Swedish Bioinformatics Workshop, this year given in Skövde, will have a chance seeing me talk about how sequencing depth influences the picture we get of the environmental resistance gene diversity. I think the topic is very urgent and interesting, and will likely come back to it in a more thorough blog post later. There are also a few other very interesting talks, for example about metagenomic gene quantification, and en masse sequencing of E. coli and H. pylori isolates. I think all attendants are in for a treat! See you there!
Metaxa in Encyclopedia of Metagenomics
A long time ago, we (Martin Eriksson, Martin Hartmann, Henrik Nilsson and me) were invited to write an overview on Metaxa for the Encyclopedia of Metagenomics. I guess the workload for pulling such a project off is huge, so there’s no surprise that it has taken a while for it to be accepted, but now it is available for consumption.
Meanwhile, Metaxa have been getting regular updates, and I hope to soon be able to show you a new major update to the software, bringing it up to the next generation of metagenomics. More on that soon.
- Bengtsson-Palme J, Hartmann M, Eriksson KM, Nilsson RH: Metaxa, overview. In:Nelson K. (Ed.) Encyclopedia of Metagenomics: SpringerReference (www.springerreference.com). Springer-Verlag Berlin Heidelberg (2013). [Link]
Metagenomics and Next Generation Sequencing Lab
Those attending the Metagenomics lab (part of the basic NGS course for PhD students given at GU this week), can find the material for the lab on this page:
https://microbiology.se/ngs-metagenomics-lab/
Of course, the page is open for anyone else as well, although you won’t get the support that the GU students are given.
An update to Megraft
You might remember that I a long time ago promised a minor update to Megraft. I then forgot about actually posting the update. So it’s very much about time, the updated 1.0.2 version of Megraft. The new thing in this version is improved handling of sequences with N’s (unknown bases) in them, and improved handling of sequences with strange sequence IDs (which sometimes have confused Megraft 1.0.1). The update can be downloaded here.
Metagenomics and the Hype Cycle
I was creating the diagram below an upcoming presentation, and I realized that the exponential growth in published metagenomics papers might be coming to an end. Interestingly enough the small drop in pace the recent years (701 -> 983 -> 1148) reminds me of the Hype Cycle, where we would (if my projection holds) have reached the “Peak of Inflated Expectations”, which means that we will see a rapid drop in the number of metagenomics publications in the next few years, as the field moves on.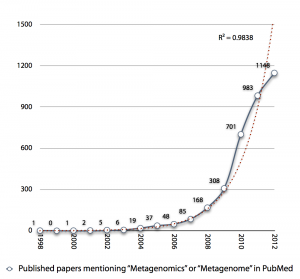
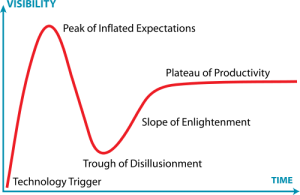 The thought is interesting, but it seems a little bit early to draw any conclusions from the number of publications, yet. It is still kind of strange to note, though, that more than 20% of metagenomics publications (740/3547) are review papers. Come on, let’s do some science first and then review it… Anyway, it’ll be interesting to see what 2013 has in store for us.
The thought is interesting, but it seems a little bit early to draw any conclusions from the number of publications, yet. It is still kind of strange to note, though, that more than 20% of metagenomics publications (740/3547) are review papers. Come on, let’s do some science first and then review it… Anyway, it’ll be interesting to see what 2013 has in store for us.
Metaxa updated to version 1.1.2
Some users have asked me to fix a table output bug in Metaxa, and I have finally got around to do so. The fix is released today in the 1.1.2 Metaxa package (download here). This version also brings an updated manual (finally), as the User’s Guide has lagged behind since version 1.0. Please continue to report bugs to metaxa [at sign] microbiology [dot] se
Megraft paper in print
I just learned from Research in Microbiology that the paper on our software Megraft has now been assigned a volume and an issue. The proper way of referencing Megraft should consequently now be:
Bengtsson J, Hartmann M, Unterseher M, Vaishampayan P, Abarenkov K, Durso L, Bik EM, Garey JR, Eriksson KM, Nilsson RH: Megraft: A software package to graft ribosomal small subunit (16S/18S) fragments onto full-length sequences for accurate species richness and sequencing depth analysis in pyrosequencing-length metagenomes. Research in Microbiology. Volume 163, Issues 6–7 (2012), 407–412, doi: 10.1016/j.resmic.2012.07.001. [Paper link]
Megraft is currently at version 1.0.1, but I have a slightly updated version in the pipeline which will be made available later this fall.