Talk tomorrow at Swedish Bioinformatics Workshop
Those of you attending the Swedish Bioinformatics Workshop, this year given in Skövde, will have a chance seeing me talk about how sequencing depth influences the picture we get of the environmental resistance gene diversity. I think the topic is very urgent and interesting, and will likely come back to it in a more thorough blog post later. There are also a few other very interesting talks, for example about metagenomic gene quantification, and en masse sequencing of E. coli and H. pylori isolates. I think all attendants are in for a treat! See you there!
A poor excuse…
I feel very sorry that I have been a little bit unresponsive for the last couple of weeks. I have received several questions regarding the PETKit and ITSx that i have not yet got around to answer. I am very sorry for that inconvenience. The reason (not a good excuse, but still) is that I have been overloaded with grant applications. This will continue through the rest of september, so please be patient until October if I don’t reply e-mails. If you need a quick response, please state so very clearly, and I might be able to squeeze you in before the start of October. Otherwise, see you at the other end of the tunnel! Thanks for the understanding.
Our new home
Last Friday, our research group moved into our new facilities at the Department of Infectious Diseases. I am very happy with my new room and my new view, both depicted below.
Our new affiliation is:
Department of Infectious Diseases, Institute of Biomedicine, Sahlgrenska Academy, University of Gothenburg
Regarding ResearchGate and paper requests
I have recently started to receive requests for full-text versions of my publications on ResearchGate. That’s great, but I have yet to figure out how to send them over, without breaking any agreements. As I am in a somewhat intensive work-period at the moment, please forgive me for not spending time on ResearchGate right now. And if you would like full-text versions of my publications, please send me an e-mail! I’ll be glad to help!
Published paper: ITSx
The paper describing our software tool ITSx has now gone online as an Early View paper on the Methods in Ecology and Evolution website. The software just recently left its beta-status behind, and with the paper out as well, we hope that as many people as possible will find use for the software in barcoding efforts of the ITS region. If you’re not familiar with the software – or its predecessor; the fungal ITS Extractor – here is a brief description of what it does:
ITSx is a Perl-based software tool that extracts the ITS1, 5.8S and ITS2 sequences – as well as full-length ITS sequences – from high-throughput sequencing data sets. To achieve this, we use carefully crafted hidden Markov models (HMMs), computed from large alignments of a total of 20 groups of eukaryotes. Testing has shown that ITSx has close to 100% detection accuracy, and virtually zero false-positive extractions. Additionally, it supports multiple processor cores, and is therefore suitable for running also on very large datasets. It is also able to eliminate non-ITS sequences from a given input dataset.
While ITSx supports extractions of ITS sequences from at least 20 different eukaryotic lineages, we ourselves have considerably less experience with many of the eukaryote groups outside of the fungi. We therefore release ITSx with the intent that the research community will evaluate its performance also in other parts of the eukaryote tree, and if necessary contribute data required to address also those lineages in a thorough way.
The ITSx paper can at the moment be cited as:
Bengtsson-Palme, J., Ryberg, M., Hartmann, M., Branco, S., Wang, Z., Godhe, A., De Wit, P., Sánchez-García, M., Ebersberger, I., de Sousa, F., Amend, A. S., Jumpponen, A., Unterseher, M., Kristiansson, E., Abarenkov, K., Bertrand, Y. J. K., Sanli, K., Eriksson, K. M., Vik, U., Veldre, V., Nilsson, R. H. (2013), Improved software detection and extraction of ITS1 and ITS2 from ribosomal ITS sequences of fungi and other eukaryotes for analysis of environmental sequencing data. Methods in Ecology and Evolution. doi: 10.1111/2041-210X.12073
Metaxa and HMMER 3.1b
As you might be aware, a new version of HMMER is out since late May. You might wonder how Metaxa (relying on HMMER3) will work if you update to the new version of HMMER, and I have finally got around to test it! The answer, according to my somewhat limited testing, is that Metaxa 1.1.2 seems to be working fine with HMMER 3.1.
You might need to go into the database directory (“metaxa_db”; should be located in the same directory as the Metaxa binaries), and remove all the files ending with suffixes .h3f .h3i .h3m and .h3p inside the “HMMs” directory. On most installation, this should not be necessary. Myself, I just plugged HMMER 3.1 in and started Metaxa, but if you get error messages complaining that “Error: bad format, binary auxfiles,
binary auxfiles are in an outdated HMMER format (3/b); please hmmpress your HMM file again”, then you should try removing the files and re-running Metaxa. This might especially be a problem on older Metaxa versions. [Update: Note that this fix will likely not work with ITSx!]
Bear in mind that I have not run thorough testing on Metaxa and HMMER 3.1, and probably won’t for the 1.1.2 version, since there’s a 2.0 version waiting just around the corner…
Additionally, if you experience problems with Megraft, you should try the same fix as for Metaxa, but with the Megraft database directory instead. Regarding ITSx, a minor update will be released very soon, which also will address HMMER 3.1b compatibility. [Update: See this post for how to work around HMMER 3.1 problems with ITSx.]
Happy barcoding everyone!
I have joined ResearchGate
On a side note, I just joined Research Gate (my profile). I’ve noted that it generates kind of the same kind of belonging-to-a-group feeling that registering on Facebook did way back, when co-author after co-author starts following you. Still, I haven’t figured out exactly what to use it for yet; it certainly seem more useful than academia.edu, with abilities to ask questions etc., but is anyone of you really using ResearchGate for this? Or is it rather just another showcasing window for researchers (much like my Publications page)? Please feel free do add your opinions as comments to this post!
Server upgrades
I’ve been informed by my web service provider that there will potentially be downtime of this site on the 13th of February (Wednesday this week), due to a server upgrade. I hope this will cause as little trouble as possible (both for you and for me).
Metagenomics and the Hype Cycle
I was creating the diagram below an upcoming presentation, and I realized that the exponential growth in published metagenomics papers might be coming to an end. Interestingly enough the small drop in pace the recent years (701 -> 983 -> 1148) reminds me of the Hype Cycle, where we would (if my projection holds) have reached the “Peak of Inflated Expectations”, which means that we will see a rapid drop in the number of metagenomics publications in the next few years, as the field moves on.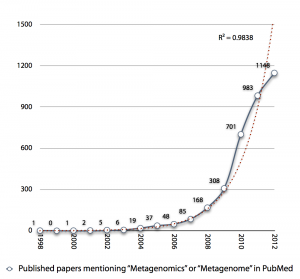
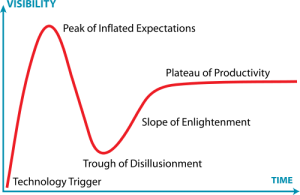 The thought is interesting, but it seems a little bit early to draw any conclusions from the number of publications, yet. It is still kind of strange to note, though, that more than 20% of metagenomics publications (740/3547) are review papers. Come on, let’s do some science first and then review it… Anyway, it’ll be interesting to see what 2013 has in store for us.
The thought is interesting, but it seems a little bit early to draw any conclusions from the number of publications, yet. It is still kind of strange to note, though, that more than 20% of metagenomics publications (740/3547) are review papers. Come on, let’s do some science first and then review it… Anyway, it’ll be interesting to see what 2013 has in store for us.
Introducing the PETKit
You know the feeling when your assembler supports paired-end sequences, but your FASTQ quality filterer doesn’t care about what pairs that belong together? Meaning that you end up with a mess of sequences that you have to script together in some way. Gosh, that feeling is way too common. It is for situations like that I have put together the Paired-End ToolKit (PETKit), a collection of FASTQ/FASTA sequence handling programs written in Perl. Currently the toolkit contains three command-line tools that does sequence conversion, quality filtering, and ORF prediction, all adapted for paired-end sequences specifically. You can read more about the programs, which are released as open source software, on the PETKit page. At the moment they lack proper documentation, but running the software with the “–help” option should bring up a useful set of options for each tool. This is still considered beta-software, so any bug reports, and especially suggestions, are welcome.
Also, if you have an idea of another problem that is unsolved or badly executed for paired-end sequences, let me know, and I will see if I can implement it in PETKit.

