New beta brings major Metaxa2 updates
I am very happy to announce that a first public beta version of Metaxa2 version 2.2 has been released today! This new version brings two big and a number of small improvements to the Metaxa2 software (1). The first major addition is the introduction of the Metaxa2 Database Builder, which allows the user to create custom databases for virtually any genetic barcoding region. The second addition, which is related to the first, is that the classifier has been rewritten to have a more solid mathematical foundation. I have been promising that these updates were coming “soon” for one and a half years, but finally the end-product is good enough to see some real world testing. Bear in mind though that this is still a beta version that could contain obscure bugs. Here follows a list of new features (with further elaboration on a few below):
- The Metaxa2 Database Builder
- Support for additional barcoding genes, virtually any genetic region can now be used for taxonomic classification in Metaxa2
- The Metaxa2 database repository, which can be accessed through the new metaxa2_install_database tool
- Improved classification scoring model for better clarity and sensitivity
- A bundled COI database for athropods, showing off the capabilities of the database builder
- Support for compressed input files (gzip, zip, bzip, dsrc)
- Support for auto-detection of database locations
- Added output of probable taxonomic origin for sequences with reliability scores at each rank, made possible by the updated classifier
- Added the -x option for running only the extraction without the classification step
- Improved memory handling for very large rRNA datasets in the classifier (millions of sequences)
- This update also fixes a bug in the metaxa2_rf tool that could cause bias in very skewed datasets with small numbers of taxa
The new version of Metaxa2 can be downloaded here, and for those interested I will spend the rest of this post outlining the Metaxa2 Database Builder. The information below is also available in a slightly extended version in the software manual.
The major enhancement in Metaxa2 version 2.2 is the ability to use custom databases for classification. This means that the user can now make their own database for their own barcoding region of choice, or download additional databases from the Metaxa2 Database Repository. The selection of other databases is made through the “-g” option already existing in Metaxa2. As part of these changes, we have also updated the classification scoring model for better stringency and sensitivity across multiple databases and different genes. The old scoring system can still be used by specifying the –scoring_model option to “old”.
There are two different main operating modes of the Metaxa2 Database Builder, as well as a hybrid mode combining the features of the two other modes. The divergent and conserved modes work in almost completely different ways and deal with two different types of barcoding regions. The divergent mode is designed to deal with barcoding regions that exhibit fairly large variation between taxa within the same taxonomic domain. Such regions include, e.g., the eukaryotic ITS region, or the trnL gene used for plant barcoding. In the other mode – the conserved mode – a highly conserved barcoding region is expected (at least within the different taxonomic domains). Genes that fall into this category would be, e.g., the 16S SSU rRNA, and the bacterial rpoB gene. This option would most likely also be suitable for barcoding within certain groups of e.g. plants, where similarity of the barcoding regions can be expected to be high. There is also a third mode – the hybrid mode – that incorporates features of both the other. The hybrid mode is more experimental in nature, but could be useful in situations where both the other modes perform poorer than desired.
In the divergent (default) mode, the database builder will start by clustering the input sequences at 20% identity using USEARCH (2). All clusters generated from this process are then individually aligned using MAFFT (3). Those alignments are split into two regions, which are used to build two hidden Markov models for each cluster of sequences. These models will be less precise, but more sensitive than those generated in the conserved mode. In the divergent mode, the database builder will attempt to extract full-length sequences from the input data, but this may be less successful than in the conserved mode.
In the conserved mode, on the other hand, the database builder will first extract the barcoding region from the input sequences using models built from a reference sequence provided (see above) and the Metaxa2 extractor (1). It will then align all the extracted sequences using MAFFT and determine the conservation of each position in the alignment. When the criteria for degree of conservation are met, all conserved regions are extracted individually and are then re-aligned separately using MAFFT. The re-aligned sequences are used to build hidden Markov models representing the conserved regions with HMMER (4). In this mode, the classification database will only consist of the extracted full-length sequences.
In the hybrid mode, finally, the database builder will cluster the input sequences at 20% identity using USEARCH, and then proceed with the conserved mode approach on each cluster separately .
The actual taxonomic classification in Metaxa2 is done using a sequence database. It was shown in the original Metaxa2 paper that replacing the built-in database with a generic non-processed one was detrimental to performance in terms of accuracy (1). In the database builder, we have tried to incorporate some of the aspects of the manual database curation we did for the built-in database that can be automated. By default, all these filtration steps are turned off, but enabling them might drastically increase the accuracy of classifications based on the database.
To assess the accuracy of the constructed database, the Metaxa2 Database Builder allows for testing the detection ability and classification accuracy of the constructed database. This is done by sub-dividing the database sequences into subsets and rebuilding the database using a smaller (by default 90%), randomly selected, set of the sequence data (5). The remaining sequences (10% by default) are then classified using Metaxa2 with the subset database. The number of detections, and the numbers of correctly or incorrectly classified entries are recorded and averaged over a number of iterations (10 by default). This allows for obtaining a picture of the lower end of the accuracy of the database. However, since the evaluation only uses a subset of all sequences included in the full database, the performance of the full database actually constructed is likely to be slightly better. The evaluation can be turned on using the “–evaluate T” option.
Metaxa2 2.2 also introduces the database repository, from which the user can download additional databases for Metaxa2. To download new databases from the repository, the metaxa2_install_database command is used. This is a simple piece of software but requires internet access to function.
References
- Bengtsson-Palme J, Hartmann M, Eriksson KM, Pal C, Thorell K, Larsson DGJ, Nilsson RH: Metaxa2: Improved Identification and Taxonomic Classification of Small and Large Subunit rRNA in Metagenomic Data. Molecular Ecology Resources (2015). doi: 10.1111/1755-0998.12399 [Paper link]
- Edgar RC: Search and clustering orders of magnitude faster than BLAST. Bioinformatics, 26, 2460–2461 (2010).
- Katoh K, Standley DM: MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molecular Biology and Evolution, 30, 772–780 (2013).
- Eddy SR: Accelerated profile HMM searches. PLoS Computational Biology, 7, e1002195 (2011).
- Richardson RT, Bengtsson-Palme J, Johnson RM: Evaluating and Optimizing the Performance of Software Commonly Used for the Taxonomic Classification of DNA Sequence Data. Molecular Ecology Resources, 17, 4, 760–769 (2017). doi: 10.1111/1755-0998.12628
Published paper: Investigating resistomes using metagenomics
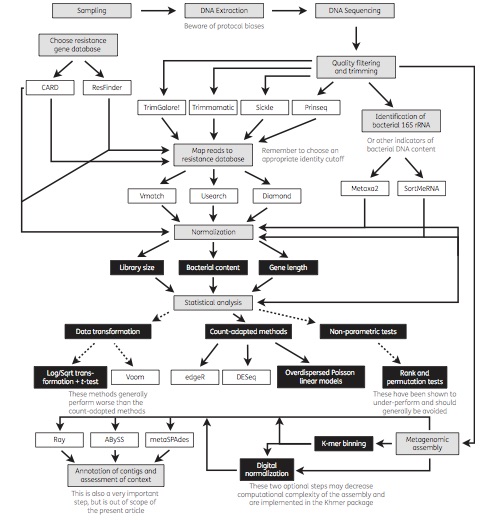
Today, a review paper which I wrote together with Joakim Larsson and Erik Kristiansson was published in Journal of Antimicrobial Chemotherapy (1). We have for a long time used metagenomic DNA sequencing to study antibiotic resistance in different environments (2-6), including in the human microbiota (7). Generally, our ultimate purpose has been to assess the risks to human health associated with resistance genes in the environment. However, a multitude of methods exist for metagenomic data analysis, and over the years we have learned that not all methods are suitable for the investigation of resistance genes for this purpose. In our review paper, we describe and discuss current methods for sequence handling, mapping to databases of resistance genes, statistical analysis and metagenomic assembly. We also provide an overview of important considerations related to the analysis of resistance genes, and end by recommending some of the currently used tools, databases and methods that are best equipped to inform research and clinical practice related to antibiotic resistance (see the figure from the paper below). We hope that the paper will be useful to researchers and clinicians interested in using metagenomic sequencing to better understand the resistance genes present in environmental and human-associated microbial communities.
References
- Bengtsson-Palme J, Larsson DGJ, Kristiansson E: Using metagenomics to investigate human and environmental resistomes. Journal of Antimicrobial Chemotherapy, advance access (2017). doi: 10.1093/jac/dkx199 [Paper link]
- Bengtsson-Palme J, Boulund F, Fick J, Kristiansson E, Larsson DGJ: Shotgun metagenomics reveals a wide array of antibiotic resistance genes and mobile elements in a polluted lake in India. Frontiers in Microbiology, 5, 648 (2014). doi: 10.3389/fmicb.2014.00648 [Paper link]
- Lundström S, Östman M, Bengtsson-Palme J, Rutgersson C, Thoudal M, Sircar T, Blanck H, Eriksson KM, Tysklind M, Flach C-F, Larsson DGJ: Minimal selective concentrations of tetracycline in complex aquatic bacterial biofilms. Science of the Total Environment, 553, 587–595 (2016). doi: 10.1016/j.scitotenv.2016.02.103 [Paper link]
- Bengtsson-Palme J, Hammarén R, Pal C, Östman M, Björlenius B, Flach C-F, Kristiansson E, Fick J, Tysklind M, Larsson DGJ: Elucidating selection processes for antibiotic resistance in sewage treatment plants using metagenomics. Science of the Total Environment, 572, 697–712 (2016). doi: 10.1016/j.scitotenv.2016.06.228 [Paper link]
- Pal C, Bengtsson-Palme J, Kristiansson E, Larsson DGJ: The structure and diversity of human, animal and environmental resistomes. Microbiome, 4, 54 (2016). doi: 10.1186/s40168-016-0199-5 [Paper link]
- Flach C-F, Pal C, Svensson CJ, Kristiansson E, Östman M, Bengtsson-Palme J, Tysklind M, Larsson DGJ: Does antifouling paint select for antibiotic resistance? Science of the Total Environment, 590–591, 461–468 (2017). doi: 10.1016/j.scitotenv.2017.01.213 [Paper link]
- Bengtsson-Palme J, Angelin M, Huss M, Kjellqvist S, Kristiansson E, Palmgren H, Larsson DGJ, Johansson A: The human gut microbiome as a transporter of antibiotic resistance genes between continents. Antimicrobial Agents and Chemotherapy, 59, 10, 6551–6560 (2015). doi: 10.1128/AAC.00933-15 [Paper link]
Database quality paper in special issue
I just want to highlight that the paper on strategies to improve database accuracy and usability we recently published in Proteomics (1) has been included in their most recent issue, which is a special issue focusing on Data Quality Issues in Proteomics. I highly recommend reading our paper (of course) and many of the other in the special issue. Happy reading!
On another note, I will be giving a talk next Wednesday (October 5th) on a seminar day on next generation sequencing in clinical microbiology, titled “Antibiotic resistance in the clinic and the environment – There and back again“. You are very welcome to the lecture hall at floor 3 in our building at Guldhedsgatan 10A here in Gothenburg if you are interested! (Bear in mind though that it all starts at 8.15 in the morning.)
Finally, it seems that I am going to the Next Generation Sequencing Congress in London this year, which will be very fun! Hope to see some of you dealing with sequencing there!
References
- Bengtsson-Palme J, Boulund F, Edström R, Feizi A, Johnning A, Jonsson VA, Karlsson FH, Pal C, Pereira MB, Rehammar A, Sánchez J, Sanli K, Thorell K: Strategies to improve usability and preserve accuracy in biological sequence databases. Proteomics, 16, 18, 2454–2460 (2016). doi: 10.1002/pmic.201600034 [Paper link]
Published paper: Annotating fungi from the built environment
MycoKeys today put a paper online which I was involved in. The paper describes the results of a workshop in May, when we added and refined annotations for fungal ITS sequences according to the MIxS-Built Environment annotation standard (1). Fungi have been associated with a range of unwanted effects in the built environment, including asthma, decay of building materials, and food spoilage. However, the state of the metadata annotation of fungal DNA sequences from the built environment is very much incomplete in public databases. The workshop aimed to ease a little part of this problem, by distributing the re-annotation of public fungal ITS sequences across 36 persons. In total, we added or changed of 45,488 data points drawing from published literature, including addition of 8,430 instances of countries of collection, 5,801 instances of building types, and 3,876 instances of surface-air contaminants. The results have been implemented in the UNITE database and shared with other online resources. I believe, that distributed initiatives like this (and the ones I have been involved in in the past (2,3)) serve a very important purpose for establishing better annotation of sequence data, an issue I have brought up also for sequences outside of barcoding genes (4). The full paper can be found here.
References
- Abarenkov K, Adams RI, Laszlo I, Agan A, Ambrioso E, Antonelli A, Bahram M, Bengtsson-Palme J, Bok G, Cangren P, Coimbra V, Coleine C, Gustafsson C, He J, Hofmann T, Kristiansson E, Larsson E, Larsson T, Liu Y, Martinsson S, Meyer W, Panova M, Pombubpa N, Ritter C, Ryberg M, Svantesson S, Scharn R, Svensson O, Töpel M, Untersehrer M, Visagie C, Wurzbacher C, Taylor AFS, Kõljalg U, Schriml L, Nilsson RH: Annotating public fungal ITS sequences from the built environment according to the MIxS-Built Environment standard – a report from a May 23-24, 2016 workshop (Gothenburg, Sweden). MycoKeys, 16, 1–15 (2016). doi: 10.3897/mycokeys.16.10000
- Kõljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M, Bates ST, Bruns TT, Bengtsson-Palme J, Callaghan TM, Douglas B, Drenkhan T, Eberhardt U, Dueñas M, Grebenc T, Griffith GW, Hartmann M, Kirk PM, Kohout P, Larsson E, Lindahl BD, Lücking R, Martín MP, Matheny PB, Nguyen NH, Niskanen T, Oja J, Peay KG, Peintner U, Peterson M, Põldmaa K, Saag L, Saar I, Schüßler A, Senés C, Smith ME, Suija A, Taylor DE, Telleria MT, Weiß M, Larsson KH: Towards a unified paradigm for sequence-based identification of Fungi. Molecular Ecology, 22, 21, 5271–5277 (2013). doi: 10.1111/mec.12481
- Nilsson RH, Hyde KD, Pawlowska J, Ryberg M, Tedersoo L, Aas AB, Alias SA, Alves A, Anderson CL, Antonelli A, Arnold AE, Bahnmann B, Bahram M, Bengtsson-Palme J, Berlin A, Branco S, Chomnunti P, Dissanayake A, Drenkhan R, Friberg H, Frøslev TG, Halwachs B, Hartmann M, Henricot B, Jayawardena R, Jumpponen A, Kauserud H, Koskela S, Kulik T, Liimatainen K, Lindahl B, Lindner D, Liu J-K, Maharachchikumbura S, Manamgoda D, Martinsson S, Neves MA, Niskanen T, Nylinder S, Pereira OL, Pinho DB, Porter TM, Queloz V, Riit T, Sanchez-García M, de Sousa F, Stefaczyk E, Tadych M, Takamatsu S, Tian Q, Udayanga D, Unterseher M, Wang Z, Wikee S, Yan J, Larsson E, Larsson K-H, Kõljalg U, Abarenkov K: Improving ITS sequence data for identification of plant pathogenic fungi. Fungal Diversity, 67, 1, 11–19 (2014). doi: 10.1007/s13225-014-0291-8
- Bengtsson-Palme J, Boulund F, Edström R, Feizi A, Johnning A, Jonsson VA, Karlsson FH, Pal C, Pereira MB, Rehammar A, Sánchez J, Sanli K, Thorell K: Strategies to improve usability and preserve accuracy in biological sequence databases. Proteomics, Early view (2016). doi: 10.1002/pmic.201600034
Published paper: Strategies for better databases
I am happy to announce that our Viewpoint article on strategies for improving sequence databases has now been published in the journal Proteomics. The paper (1) defines some central problems hampering genomic, proteomic and metagenomic analyses and suggests five strategies to improve the situation:
- Clearly separate experimentally verified and unverified sequence entries
- Enable a system for tracing the origins of annotations
- Separate entries with high-quality, informative annotation from less useful ones
- Integrate automated quality-control software whenever such tools exist
- Facilitate post-submission editing of annotations and metadata associated with sequences
The paper is not long, so I encourage you to read it in its entirety. We believe that spreading this knowledge and pushing solutions to problems related to poor annotation metadata is vastly important in this era of big data. Although we specifically address protein-coding genes in this paper, the same logic also applies to other types of biological sequences. In this way the paper is related to my previous work with Henrik Nilsson on improving annotation data for taxonomic barcoding genes (2-4). This paper was one of the main end-results of the GoBiG network, and the backstory on the paper follows below the references…
References
- Bengtsson-Palme J, Boulund F, Edström R, Feizi A, Johnning A, Jonsson VA, Karlsson FH, Pal C, Pereira MB, Rehammar A, Sánchez J, Sanli K, Thorell K: Strategies to improve usability and preserve accuracy in biological sequence databases. Proteomics, Early view (2016). doi: 10.1002/pmic.201600034
- Kõljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M, Bates ST, Bruns TT, Bengtsson-Palme J, Callaghan TM, Douglas B, Drenkhan T, Eberhardt U, Dueñas M, Grebenc T, Griffith GW, Hartmann M, Kirk PM, Kohout P, Larsson E, Lindahl BD, Lücking R, Martín MP, Matheny PB, Nguyen NH, Niskanen T, Oja J, Peay KG, Peintner U, Peterson M, Põldmaa K, Saag L, Saar I, Schüßler A, Senés C, Smith ME, Suija A, Taylor DE, Telleria MT, Weiß M, Larsson KH: Towards a unified paradigm for sequence-based identification of Fungi. Molecular Ecology, 22, 21, 5271–5277 (2013). doi: 10.1111/mec.12481
- Nilsson RH, Hyde KD, Pawlowska J, Ryberg M, Tedersoo L, Aas AB, Alias SA, Alves A, Anderson CL, Antonelli A, Arnold AE, Bahnmann B, Bahram M, Bengtsson-Palme J, Berlin A, Branco S, Chomnunti P, Dissanayake A, Drenkhan R, Friberg H, Frøslev TG, Halwachs B, Hartmann M, Henricot B, Jayawardena R, Jumpponen A, Kauserud H, Koskela S, Kulik T, Liimatainen K, Lindahl B, Lindner D, Liu J-K, Maharachchikumbura S, Manamgoda D, Martinsson S, Neves MA, Niskanen T, Nylinder S, Pereira OL, Pinho DB, Porter TM, Queloz V, Riit T, Sanchez-García M, de Sousa F, Stefaczyk E, Tadych M, Takamatsu S, Tian Q, Udayanga D, Unterseher M, Wang Z, Wikee S, Yan J, Larsson E, Larsson K-H, Kõljalg U, Abarenkov K: Improving ITS sequence data for identification of plant pathogenic fungi. Fungal Diversity, 67, 1, 11–19 (2014). doi: 10.1007/s13225-014-0291-8
- Nilsson RH, Tedersoo L, Ryberg M, Kristiansson E, Hartmann M, Unterseher M, Porter TM, Bengtsson-Palme J, Walker D, de Sousa F, Gamper HA, Larsson E, Larsson K-H, Kõljalg U, Edgar R, Abarenkov K: A comprehensive, automatically updated fungal ITS sequence dataset for reference-based chimera control in environmental sequencing efforts. Microbes and Environments, 30, 2, 145–150 (2015). doi: 10.1264/jsme2.ME14121
Backstory
In June 2013, the Gothenburg Bioinformatics Group for junior scientists (GoBiG) arranged a workshop with two themes: “Parallelized quantification of genes in large metagenomic datasets” and “Assigning functional predictions to NGS data”. The following discussion on how to database quality influenced results and what could be done to improve the situation was rather intense, and several good ideas were thrown around. I took notes from the meeting, and in the evening I put them down during a warm summer night at the balcony. In fact, the notes were good enough to be an early embryo for a manuscript. So I sent it to some of the most active GoBiG members (Kaisa Thorell and Fredrik Boulund), who were positive regarding the idea to turn it into a manuscript. I wrote it together more properly and we decided that everyone who contributed with ideas at the meeting would be invited to become co-authors. We submitted the manuscript in early 2014, only to see it (rather brutally) rejected. At that point most of us were sucked up in their own projects, so nothing happened to this manuscript for over a year. Then we decided to give it another go, updated the manuscript heavily and changed a few parts to better reflect the current database situation (at this point, e.g., UniProt had already started implementing some of our suggested ideas). Still, some of the proposed strategies were more radical in 2013 than they would be now, more than three years later. We asked the Proteomics editors if they would be interested in the manuscript, and they turned out to be very positive. Indeed, the entire experience with the editors at Proteomics has been very pleasant. I am very thankful to the GoBiG team for this time, and to the editors at Proteomics who saw the value of this manuscript.
Talk at Swedish Bioinformatics Workshop
I have had the pleasure to be chosen as a speaker for next week’s (ten days from now) Swedish Bioinformatics Workshop. My talk is entitled “Turn up the signal – wipe out the noise: Gaining insights into bacterial community functions using metagenomic data“, and will largely deal with the same questions as my talk on EDAR3 in May this year. As then, the talk will highlight the some particular pitfalls related to interpretation of data, and exemplify how flawed analysis practices can result in misleading conclusions regarding community function, and use examples from our studies of environments subjected to pharmaceutical pollution in India, the effect of travel on the human resistome, and modern municipal wastewater treatment processes.
The talk will take place on Thursday, September 24, 2015 at 16:30. The full program for the conference can be found here. And also, if you want a sneak peak of the talk, you can drop by on Friday 13.00 at Chemistry and Molecular Biology, where I will give a seminar on the same topic in the Monthly Bioinformatic Practical Meetings series.
Talk on the EDAR2015 conference
I will be giving a talk at the Third International symposium on the environmental dimension of antibiotic resistance (EDAR2015) next month (five weeks from now. The talk is entitled “Turn up the signal – wipe out the noise: Gaining insights into antibiotic resistance of bacterial communities using metagenomic data“, and will deal with handling of metagenomic data in antibiotic resistance gene research. The talk will highlight the some particular pitfalls related to interpretation of data, and exemplify how flawed analysis practices can result in misleading conclusions regarding antibiotic resistance risks. I will particularly address how taxonomic composition influences the frequencies of resistance genes, the importance of knowledge of the functions of the genes in the databases used, and how normalization strategies influence the results. Furthermore, we will show how the context of resistance genes can allow inference of their potential to spread to human pathogens from environmental or commensal bacteria. All these aspects will be exemplified by data from our studies of environments subjected to pharmaceutical pollution in India, the effect of travel on the human resistome, and modern municipal wastewater treatment processes.
The talk will take place on Monday, May 18, 2015 at 13:20. The full scientific program for the conference can be found here. Registration for the conference is still possible, although not for the early-bird price. I look forward to see a lot of the people who will attend the conference, and hopefully also you!
Published paper: ITS chimera dataset
A couple of days ago, a paper I have co-authored describing an ITS sequence dataset for chimera control in fungi went online as an advance online publication in Microbes and Environments. There are several software tools available for chimera detection (e.g. Henrik Nilsson‘s fungal chimera checker (1) and UCHIME (2)), but these generally rely on the presence of a chimera-free reference dataset. Until now, there was no such dataset is for the fungal ITS region, and we in this paper (3) introduce a comprehensive, automatically updated reference dataset for fungal ITS sequences based on the UNITE database (4). This dataset supports chimera detection throughout the fungal kingdom and for full-length ITS sequences as well as partial (ITS1 or ITS2 only) datasets. We estimated the dataset performance on a large set of artificial chimeras to be above 99.5%, and also used the dataset to remove nearly 1,000 chimeric fungal ITS sequences from the UNITE database. The dataset can be downloaded from the UNITE repository. Thereby, it is also possible for users to curate the dataset in the future through the UNITE interactive editing tools.
References:
- Nilsson RH, Abarenkov K, Veldre V, Nylinder S, Wit P de, Brosché S, Alfredsson JF, Ryberg M, Kristiansson E: An open source chimera checker for the fungal ITS region. Molecular Ecology Resources, 10, 1076–1081 (2010).
- Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics, 27, 16, 2194-2200 (2011). doi:10.1093/bioinformatics/btr381
- Nilsson RH, Tedersoo L, Ryberg M, Kristiansson E, Hartmann M, Unterseher M, Porter TM, Bengtsson-Palme J, Walker D, de Sousa F, Gamper HA, Larsson E, Larsson K-H, Kõljalg U, Edgar R, Abarenkov K: A comprehensive, automatically updated fungal ITS sequence dataset for reference-based chimera control in environmental sequencing efforts. Microbes and Environments, Advance Online Publication (2015). doi: 10.1264/jsme2.ME14121
- Kõljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M, Bates ST, Bruns TT, Bengtsson-Palme J, Callaghan TM, Douglas B, Drenkhan T, Eberhardt U, Dueñas M, Grebenc T, Griffith GW, Hartmann M, Kirk PM, Kohout P, Larsson E, Lindahl BD, Lücking R, Martín MP, Matheny PB, Nguyen NH, Niskanen T, Oja J, Peay KG, Peintner U, Peterson M, Põldmaa K, Saag L, Saar I, Schüßler A, Senés C, Smith ME, Suija A, Taylor DE, Telleria MT, Weiß M, Larsson KH: Towards a unified paradigm for sequence-based identification of Fungi. Molecular Ecology, 22, 21, 5271–5277 (2013). doi: 10.1111/mec.12481
Scientific Data – a way of getting credit for data
In an interesting development, Nature Publishing Group has launched a new initiative: Scientific Data – a online-only open access journal that publishes data sets without the demand of testing scientific hypotheses in connection to the data. That is, the data itself is seen as the valuable product, not any findings that might result from it. There is an immediate upside of this; large scientific data sets might be accessible to the research community in a way that enables proper credit for the sample collection effort. Since there is no demand for a full analysis of the data, the data itself might quicker be of use to others, without worrying that someone else might steal the bang of the data per se. I also see a possible downside, though. It would be easy to hold on to the data until you have analyzed it yourself, and then release it separately just about when you submit the paper on the analysis, generating extra papers and citation counts. I don’t know if this is necessarily bad, but it seems it could contribute to “publishing unit dilution”. Nevertheless, I believe that this is overall a good initiative, although how well it actually works will be up to us – the scientific community. Some info copied from the journal website:
Scientific Data’s main article-type is the Data Descriptor: peer-reviewed, scientific publications that provide an in-depth look at research datasets. Data Descriptors are a combination of traditional scientific publication content and structured information curated in-house, and are designed to maximize reuse and enable searching, linking and data mining. (…) Scientific Data aims to address the increasing need to make research data more available, citable, discoverable, interpretable, reusable and reproducible. We understand that wider data-sharing requires credit mechanisms that reward scientists for releasing their data, and peer evaluation mechanisms that account for data quality and ensure alignment with community standards.
Published paper: BacMet Database
It seems like our paper on the recently launched database on resistance genes against antibacterial biocides and metals (BacMet) has gone online as an advance access paper in Nucleic Acids Research today. Chandan Pal – the first author of the paper, and one of my close colleagues as well as my roommate at work – has made a tremendous job taking the database from a list of genes and references, to a full-fledged browsable and searchable database with a really nice interface. I have contributed along the process, and wrote the lion’s share of the code for the BacMet-Scan tool that can be downloaded along with the database files.
BacMet is a curated source of bacterial resistance genes against antibacterial biocides and metals. All gene entries included have at least one experimentally confirmed resistance gene with references in scientific literature. However, we have also made a homology-based prediction of genes that are likely to share the same resistance function (the BacMet predicted dataset). We believe that the BacMet database will make it possible to better understand co- and cross-resistance of biocides and metals to antibiotics within bacterial genomes and in complex microbial communities from different environments.
The database can be easily accessed here: http://bacmet.biomedicine.gu.se, and use of the database in scientific work can cite the following paper, which recently appeared in Nucleic Acids Research:
Pal C, Bengtsson-Palme J, Rensing C, Kristiansson E, Larsson DGJ: BacMet: Antibacterial Biocide and Metal Resistance Genes Database. Nucleic Acids Research. Database issue, advance access. doi: 10.1093/nar/gkt1252 [Paper link]